 The calomel electrode is a reference electrode, especially in older publications. Mercurous chloride is employed extensively in electrochemistry, taking advantage of the ease of its oxidation and reduction reactions. Main article: Saturated calomel electrode Hg 2Cl 2 + 2 NH 3 → Hg + Hg(NH 2)Cl + NH 4Cl Calomel electrode 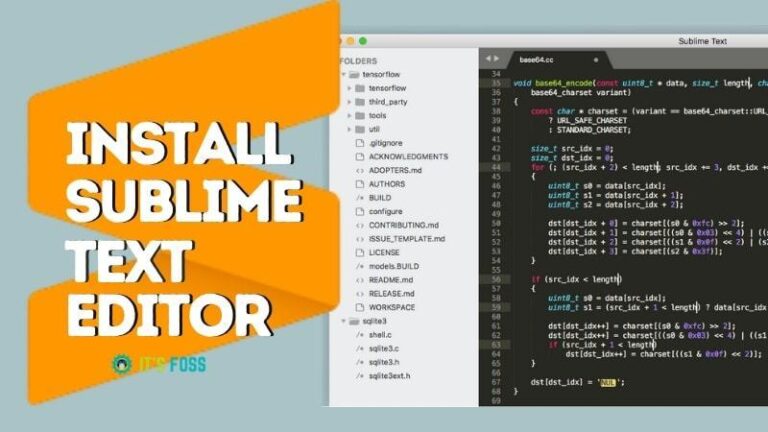
It can be prepared via metathesis reaction involving aqueous mercury(I) nitrate using various chloride sources including NaCl or HCl.Ģ HCl + Hg 2(NO 3) 2 → Hg 2Cl 2 + 2 HNO 3Īmmonia causes Hg 2Cl 2 to disproportionate: Mercurous chloride forms by the reaction of elemental mercury and mercuric chloride: The overall coordination of each Hg atom is octahedral as, in addition to the two nearest neighbours, there are four other Cl atoms at 321 pm. The Hg–Hg bond length of 253 pm (Hg–Hg in the metal is 300 pm) and the Hg–Cl bond length in the linear Hg 2Cl 2 unit is 243 pm. The unit cell of the crystal structure is shown below: The mineral calomel crystallizes in the tetragonal system, with space group I4/m 2/m 2/m. Mercury is unique among the group 12 metals for its ability to form the M–M bond so readily. 
Researchers used that same mercury, found deep in latrine pits, to retrace the locations of their respective locations and campsites. Lewis and Clark brought calomel on their expedition. Yellow fever was also treated with calomel. Calomel was given to patients as a purgative or cathartic until they began to salivate and was often administered to patients in such great quantities that their hair and teeth fell out. Benjamin Rush was a well-known advocate of mercury in medicine and used calomel to treat sufferers of yellow fever during its outbreak in Philadelphia in 1793. It was prescribed by doctors in America throughout the 18th century, and during the revolution, to make patients regurgitate and release their body from "impurities". Mercury became a popular remedy for a variety of physical and mental ailments during the age of " heroic medicine". Until fairly recently, it was also used as a horticultural fungicide, most notably as a root dip to help prevent the occurrence of clubroot amongst crops of the family Brassicaceae. Ĭalomel was taken internally and used as a laxative, for example to treat George III in 1801, and disinfectant, as well as in the treatment of syphilis, until the early 20th century. It is also referred to as the mineral horn quicksilver or horn mercury. 
The "black" name (somewhat surprising for a white compound) is probably due to its characteristic disproportionation reaction with ammonia, which gives a spectacular black coloration due to the finely dispersed metallic mercury formed. The name calomel is thought to come from the Greek καλός "beautiful", and μέλας "black" or καλός and μέλι "honey" from its sweet taste. It is a component of reference electrodes in electrochemistry. Also known as the mineral calomel (a rare mineral) or mercurous chloride, this dense white or yellowish-white, odorless solid is the principal example of a mercury(I) compound. Mercury(I) chloride is the chemical compound with the formula Hg 2Cl 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
